Recently, the disposable defibrillation electrode tablet independently developed and designed by Medlinket has successfully passed the registration of China National Drug Administration (NMPA).
Product Name: disposable defibrillation electrode
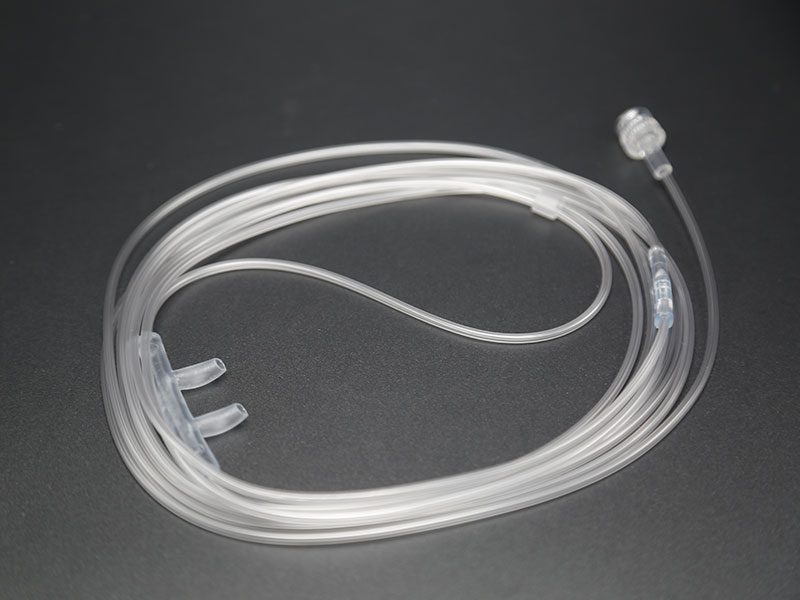
Main structure: it is composed of electrode sheet, lead wire and connector plug.
Scope of application: it can be used in external defibrillation, cardioversion and pacing.
Applicable population: patients weighing more than 25kg
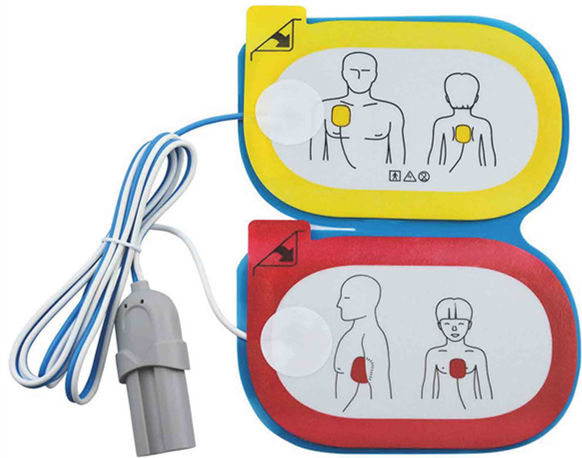
The above is the illustration of Medlinket disposable defibrillation electrode tablets. If you want to know more matching models of defibrillation electrode tablets, you can contact your sales representative at any time or send an email to sales@med -Linket.com, we will provide you with professional services.
Medlinket has always insisted on providing customers with first-class products and services, and fulfilled the mission of "making medical care easier and people healthier". Adhering to rigorous, efficient and professional services, we will work with you to promote safe, effective and compliant medical devices to the market at the fastest speed and contribute to the development of global human health.
Thank you for your support and trust!
Shenzhen Med-link Electronics Tech Co., Ltd
October 27, 2021
Previous: Medlinket's disposable NIBP cuff protector can effectively prevent cross-infection in the hospital
Next: Medlinket's Y-type multi-site SpO2 probe, a small expert in clinical home-based measurement
Related News
- Why does anesthesiology department use disposable spo2 sen...
- Medlinket's physical sign monitoring equipment is a "good...
- Medlinket won the Top 10 Best Reputation Equipment a...
- Medlinket's Infant Incubator, Warmer Temperature Probe...
- Long-term SpO2 monitoring will cause skin burn risk?
- New product recommendations:Medlinket disposable IBP ...
- Medlinket's NIBP cuff adapts to the needs of different d...
- Medlinket's Compatible Welch Allyn Smart Temp Prob...
- How to choose a reusable SpO2 sensor?
- Medlinket disposable EEG sensors to provide accurate mo...
- Medlinket's pelvic floor muscle rehabilitation probe helps...
- Pelvic floor rehabilitation probe manufacturer, manufactu...
- Why is the body cavity temperature probe generally selecte...
- Medlinket's One-Piece ECG Cable with LeadWires is ...
- Medlinket's blood oxygen probe is highly accurate, escortin...
- Medlinket professionally developed a high-precision oximet...
- Medlinket's products obtaining the UK MHRA registrat...
- Medlinket's disposable NIBP cuff protector can effective...
- Medlinket disposable defibrillation electrode are registere...
- Medlinket's Y-type multi-site SpO2 probe, a small expe...
- Medlinket’s disposable NIBP cuff, specially designed fo...
- Medlinket's disposable non-invasive EEG sensor helps to ...
- Pelvic floor rehabilitation probe manufacturer, recognize ...
- Pelvic floor rehabilitation can not be ignored ,Look for M...
- Medlinket Digital Infrared Thermometer, a good helper f...
- End expiratory carbon dioxide sensor and sampling tube acc...
- Medlinket's temp-pulse oximeter realizes five major heal...
- Low SpO2, have you found the reason behind it?
- Manufacturers of disposable EEG sensors prefer Medlinke...
- Medlinket’s home portable Temp-Pluse oximeter, scient...
- Why does anesthesiology department use disposable spo2 sen...
- Medlinket's disposable temperature probe meets the needs ...
- Medlinket Temp-plus oximeter, the patron saint of health...
- How to choose suitable disposable SpO2 sensor in differe...
- Medlinket anesthesia depth EEG sensor has obtained MHR...
- Medlinket's disposable NIBP cuff can effectively reduce ...
- The manufacturer of pelvic floor rehabilitation probe is t...
- NIBP measurement method and choice of NIBPcuff
- Medlinket's internal electrode for pelvic floor muscle the...
- Expert consensus on emergency end expiratory carbon dioxid...
- What types of oximeters are there? How to buy it?
- For ETCO2 monitoring, intubated patients are most sui...
- Medlinket's anti-jitter high-precision Temp-Pluse oxime...
- A high-precision oximeter that meets clinical testing, a
- Disposable non-invasive EEG sensor, supplied by the man...
- What are the characteristics of Medlinket’s new silicone...
- In 2021 CMEF/ICMD autumn exhibition, Medlinket invi...
- How to select spo2 sensor in various departments of the h...
- How is Medlinket's disposable non-invasive EEG sensor ...
- Internationally acclaimed oximeter——Medlinket’s tempe...
- What are the types of disposable non-invasive EEG sensor...
- To monitor the patient's respiratory status, it is necessa...
- The clinical significance of temperature management duri...
- The difference between disposable Skin-surface temperatu...
- Why should we use disposable noninvasive EEG sensors to...
- "Guardian God" for Premature Infants-ncubator Temperat...
- What is the difference between mainstream CO2 sensor a...
- The importance of disposable temperature probes in clinic...
- Application scenarios and usage methods of Disposable Sp...
- For the bidding of disposable EEG sensor manufacturers,...
- Will the SpO2 sensor cause neonatal skin burns in SpO2...
- How to choose the appropriate disposable anesthesia depth ...
- SpO2 of Novel Coronavirus Pneumonia testing standards
- Application scenarios and applicable people of different S...
- Medlinket's ETCO2 mainstream and sidestream sensors ...
- Why choose a disposable non-invasive EEG sensor adapted ...
- Medlinket's disposable non-invasive EEG sensor has been ...
- Medlinket depth-of-anesthesia sensor helps anesthesiologist...
- Medlinket Adult Finger Clip Oximetry Probe, a great h...
- FIME2018
- 2018 Med-link Medical Exhibitions Preview
- [Exhibition notice] Med-linket’s exhibition overview in ...
- 2017 American Society Of Anesthesiologists Annual Con...
- [Exhibition Invitation]Med-linket Invited You To Partic...
- Med-link Participated In The 2017 Annual Meeting Of A...
Related Products










 marketing@medxing.com
marketing@medxing.com



